Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit....
Transcript of Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit....
![Page 1: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/1.jpg)
STRUKTURA E ATOMITElektroni
![Page 2: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/2.jpg)
Gypi katodik
![Page 3: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/3.jpg)
Thompson
H x e x v = E x ev=E/H
(H x e x v) = mv2/re/m = v/Hxr
![Page 4: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/4.jpg)
Protoni
![Page 6: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/6.jpg)
Berthama e atomit
10-12cm
![Page 7: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/7.jpg)
![Page 8: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/8.jpg)
Rrezet e Rentgenit
![Page 9: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/9.jpg)
![Page 10: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/10.jpg)
Grimcasubatomike
simboli Masa (kg) shenja Ngarkesa (C)
protoni p 1.672x10-27 + 1.602x10-19
neutroni n 1.674x10-27 0 -elektroni e 9.109x10-31 - 1.602x10-19
![Page 11: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/11.jpg)
Struktura elektronike e atomit
![Page 12: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/12.jpg)
Struktura elektronike e atomit
E2 - E1 = h x υE = h x υ
![Page 13: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/13.jpg)
Numrat kuantik
Numri kuantik kryesor n = 1, 2, 3... Numri kuantik sekondar ℓ = 0 deri ℓ = n – 1per elektronin me n = 1 dhe ℓ = 0 themi se eshte ne orbitalen 1s per elektronin me n = 2 dhe ℓ = 0 ... orbitalen 2 s per elektronin me n = 2 dhe ℓ = 1 ... orbitalen 2 p per elektronin me n = 3 dhe ℓ = 2 ... orbitalen 3 d
Numri magnetikm=2l+1
Numri kuantik spinit +1/2 dhe -1/2
![Page 14: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/14.jpg)
Teoria mekaniko-valore mbi strukturen e atomit
De Broli λ=h/me x vHajzenbergu (pozita/shpejtesia)Shredingeri (ekuacioni i vales)
![Page 15: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/15.jpg)
![Page 16: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/16.jpg)
![Page 17: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/17.jpg)
![Page 18: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/18.jpg)
![Page 19: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/19.jpg)
![Page 20: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/20.jpg)
![Page 21: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/21.jpg)
![Page 22: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/22.jpg)
Struktura e molekules
![Page 23: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/23.jpg)
Lidhja kovalente
Shembuj te lidhjeve kovalente
![Page 24: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/24.jpg)
Strukturat e Luisit
![Page 25: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/25.jpg)
Shembull NF3
![Page 26: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/26.jpg)
Tre qifte elektronike tre lidhje kovalente
![Page 27: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/27.jpg)
Numri i pergjithshem i elektroneve
![Page 28: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/28.jpg)
Atomi qendror me pak elektronegativ
![Page 29: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/29.jpg)
Per tu formuar okteti te secili atom
![Page 30: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/30.jpg)
Shembull
CO3 2-
![Page 31: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/31.jpg)
![Page 32: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/32.jpg)
![Page 33: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/33.jpg)
![Page 34: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/34.jpg)
![Page 35: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/35.jpg)
![Page 36: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/36.jpg)
![Page 37: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/37.jpg)
![Page 38: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/38.jpg)
Lidhja hidrogjenore
![Page 39: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/39.jpg)
![Page 40: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/40.jpg)
Lidhja metalike
![Page 41: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/41.jpg)
![Page 42: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/42.jpg)
![Page 43: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/43.jpg)
Forcat e londonit
![Page 44: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/44.jpg)
Interaksionet dipol-dipol
![Page 45: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/45.jpg)
![Page 46: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/46.jpg)
Komponimet komplexe
Alfred Werner
![Page 47: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/47.jpg)
SHEMBUJ TE KOMPONIMEVEKOMPLEKSE
Formula molekulare
Baza e Luisit/ ligand
Acidi I Luisit
Atomidonor
NumrikoordinativI jonit
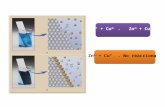
[Ag(NH3)2]+ NH3 Ag+ N 2
[Zn(CN)4]2- CN- Zn2+ C 4
[Ni(CN)4]2- CN- Ni2+ C 4
[PtCl6]2- Cl- Pt4+ Cl 6
[Ni(NH3)6]2+ NH3 Ni2+ N 6
![Page 5: Struktura e atomit · 2018. 9. 8. · Acidi I Luisit. Atomi donor: Numri koordinativ I jonit. [Ag(NH: 3) 2] + NH. 3. Ag + N. 2 [Zn(CN) 4] 2-CN-Zn. 2+ C. 4 [Ni(CN) 4] 2-CN-Ni. 2+ C.](https://reader031.fdocument.pub/reader031/viewer/2022012003/609eca4d5a2c1e418722f98f/html5/thumbnails/5.jpg)





![Elcuerpohumanotranslucido1 Cn[2]](https://static.fdocument.pub/doc/165x107/5584e740d8b42a82618b4653/elcuerpohumanotranslucido1-cn2-5585e5dd736af.jpg)







![2(NH3 4 tetraamminodiclorurocobalto(III) 2- 4...1 d2 minimizza le repulsioni fra i leganti 12 [Cr(en) 3][Ni(CN) 5] 13 [Cr(en) 3][Ni(CN) 5] 14 [Cu(bpy){NH(CH 2 CO 2) 2}] [Zn{N(CH 2](https://static.fdocument.pub/doc/165x107/603d23e995a2d97d17608292/2nh3-4-tetraamminodiclorurocobaltoiii-2-4-1-d2-minimizza-le-repulsioni-fra.jpg)



